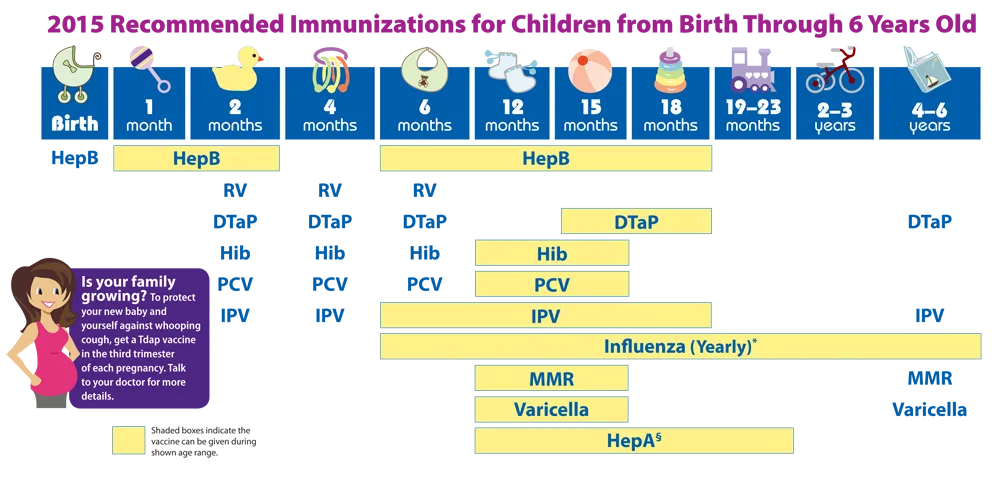
Schedule for 7-18 year olds
Vaccine schedules may be effected by manufacturing delays or backorders.
What is a Vaccine Information Statement?
A Vaccine Information Statement (VIS) is a one-page (two-sided) information sheet, produced by CDC. VISs inform vaccine recipients - or their parents or legal representatives - about the benefits and risks of a vaccine. The law requires that VISs given out whenever certain vaccinations are given.
Who must give out VISs?
All provider of vaccines, both public and private sector.
Why must VISs be used?
It is a requirement of the National Childhood Vaccine Injury Act of 1986. Their purpose is to inform vaccine recipients, or parents of children getting vaccines, about the benefits and risks of vaccines.
You can view the VIS for the vaccines we use from the list below:
- Chicken Pox Vaccine: http://www.immunize.org/vis/vis_chickenpox.asp
- HIB Vaccine: http://www.immunize.org/vis/hib99.pdf
- Hepatitis A: http://www.immunize.org/vis/v-hepa.pdf
- Hepatitis B: http://www.immunize.org/vis/hepb01.pdf
- HPV Vaccine: http://www.immunize.org/vis/hpv.pdf
- MMR Vaccine: http://www.immunize.org/vis/mmr03.pdf
- Meningitis Vaccine: http://www.immunize.org/vis/menin06.pdf
- Prevnar Vaccine: http://www.immunize.org/vis/pnPCV7.pdf
- Polio Vaccine: http://www.immunize.org/vis/ipv-00.pdf
- Rotavirus Vaccine: http://www.immunize.org/vis/rota_06.pdf
- Td Vaccine: http://www.immunize.org/vis/td2.pdf
- Tdap Vaccine: http://www.immunize.org/vis/tdap.pdf
- Inactive Influenza Vaccine: http://www.immunize.org/vis/vis_fluinactive.asp
- Live Intranasal Influenza Vaccine: http://www.immunize.org/vis/liveflu.pdf
- Other Lanuages available: http://www.immunize.org/vis/#index